Cardiovascular diseases represent the leading cause of morbidity and mortality worldwide and are responsible for 25-30% of today’s deaths worldwide. Although heart medications have been proven to be effective for the prevention and treatment of cardiac dysfunction, the research for improved therapies is ongoing, with the objective of decreasing the deaths related to cardiovascular diseases. Additionally, the potential cardiotoxic adverse side effects are one of the main causes of withdrawal of Drug candidates during Early Drug Discovery and Development.
The most common strategy for early detection of adverse cardiac symptoms of drug candidates is the use of in vitro cardiomyocyte analysis and the use of animal models. Even though the cell-based models allow for expeditious screening of cardiotoxic properties, they do not reproduce whole-organism effects. On the contrary, animal-based methods unveil a more detailed cardiotoxic profile that normally involves expensive and time-consuming assays and with many ethical concerns.
Consequently, the development of new time-effective models, with fewer ethical impediments and preserving the quality of trials is essential for a successful Drug Discovery and Development. In this scenario, the Zebrafish larvae (Danio rerio) upon 5-6 days post-fertilization (dpf) has arisen as a promising New Alternative Methodology (NAM) for the evaluation of drug-dependent cardiotoxicity and the identification of novel cardioprotective molecules. One of the strengths of the Zebrafish model is their rapid cardiac development, as by 2 dpf the cardiac is functional, and together with their transparency it allows the direct in vivo visualization of the circulatory system. As this NAM presents a high genetic homology with humans (~70% homology) and can be genetically manipulated for disease model generation, even with the expression of fluorescent proteins for direct visualization of internal structures and physiological processes, this can be applied to heart visualization.
Although apparent differences in the heart anatomy between zebrafish and humans exist, there are also vast physiological resemblances that offer an advantage as well. For instance, the speed of the heartbeat in Zebrafish ranges from 120 to 180 beats/min, which is comparable to the resting heart rate of around 70 beats/min observed in humans. In contrast, murine heart rate exceeds 300 beats/min and rat heart rate ranges from 250 to 493 beats/min.
The zebrafish heart contains two independent chambers (atrium and ventricle), and due to the existence of transgenic zebrafish models, which express Green Fluorescent Protein (GFP) in the heart, it allows easy and direct identification of the cardiotoxic effects such as arrhythmias or pericardial edemas and malformations (Figure 1) that Drug candidates may produce.
Besides, zebrafish are small, feasible to maintain, present high fecundity and fulfill high ethical standards in line with the 3Rs principle (Replacement, Reduction and Refinement of animals), reducing the number of mammals used in later phases, especially dogs.
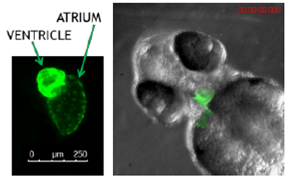
Figure 1. Zebrafish at 72 hpf expressing green fluorescent protein in the heart.
How do we perform cardiotoxicity assays at Biobide?
The Cardiotox Assay developed and validated at Biobide allows the detection of cardiac arrhythmias and cardiac malfunctions in 48 dpf zebrafish larvae, using a specific software developed in-house (cardio 4.2). By analyzing reference compounds with known cardiotoxic potential can discern between cardiotoxic and non-cardiotoxic compounds. Normally, the incubation with the test compounds lasts between 24-48 hours and at the specific endpoint times cardiac rhythm and cardiac size measurements are calculated and malformations characterized (Figure 2)
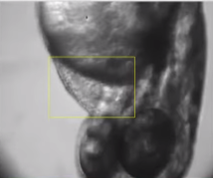
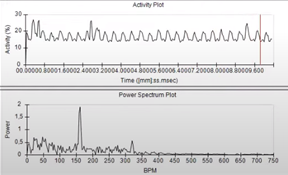
Figure 2. Assessment of cardiac rhythm in beats per minute (BPM) using Danioscope software (Nodus).
Additionally, thanks to the fluorescence of the GFP expressing cardiac cells it is feasible to identify different types of arrhythmias (including the characteristic arrhythmia 2:1, which resembles the QT prolongation in humans), and enable the possibility of deeper image analysis, as it could be the volumetric measurements of the heart chambers.
The use of Zebrafish for cardiotoxicity study is a potent NAM for the Early Drug Discovery process offering a time and cost-effective model with reliable results that fulfill the 3R principle. This is essential for the improvement of heart diseases which impact tremendously on life expectancy and quality worldwide. At Biobide we perform the Cardiotox Assay that allows the detection of cardiac arrhythmias 2:1 and cardiac phenotypic malformations in Zebrafish larvae, assuring compliance with all the quality standards and in a state-of-the-art facility.
Sources
- https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death
- D. Solomon, M.A. Pfeffer, The future of clinical trials in cardiovascular medicine, Circulation 133 (25) (2016) 2662–2670, https://doi.org/10.1161/ CIRCULATIONAHA.115.020723.
- Milani-Nejad, P.M. Janssen, Small and large animal models in cardiac contraction research: advantages and disadvantages, Pharmacol. Ther. 141 (3) (2014) 235–249, https://doi.org/10.1016/j.pharmthera.2013.10.007.
- Maciag M, Wnorowski A, Mierzejewska M, Plazinska A. Pharmacological assessment of zebrafish-based cardiotoxicity models. Biomed Pharmacother. 2022 Apr;148:112695. doi: 10.1016/j.biopha.2022.112695. Epub 2022 Feb 11. PMID: 35158142.
- T. Nguyen, Q. Lu, Y. Wang, J.N. Chen, Zebrafish as a model for cardiovascular development and disease, Drug Discov. Today Dis. Models 5 (3) (2008) 135–140, https://doi.org/10.1016/j.ddmod.2009.02.003.
- https://www.noldus.com/danioscope




