In the field of Drug Development and chemical safety assessment, evaluating the potential immunotoxicity of compounds is of utmost importance. Zebrafish has emerged as a valuable model organism for assessing immunotoxicity and Neutrophil Migration Assay offers a valuable and efficient approach.
The Neutrophil Migration Assay with zebrafish has broad applications in immunology, inflammation research, and Drug Discovery. It serves as a test for immunotoxicity evaluation and, also, enables screening potential anti-inflammatory drugs or immunomodulatory compounds to identify candidates for further development.
This article explores the potential of zebrafish Neutrophil Migration Assays as a reliable tool for evaluating immunotoxicity, advancing safety assessment strategies, and immunomodulatory properties of drug candidates in the early Drug Discovery process.
Immunotoxicity Assessment: The Need for Reliable Models
Immunotoxicity poses significant challenges in Drug Development and Safety assessment. Traditionally, the use of laboratory animals, usually rodents, has been necessary for toxicity evaluations of new Drugs in the Early Discovery Phase, being tremendously costly, time-consuming, and presenting ethical concerns. On the other hand, traditional in vitro assays have many limitations in predicting immunotoxicity. Therefore, there is a growing need for New Alternative Methods (NAMs) to overcome these problems and minimize laboratory animal use in line with the 3Rs principles (Replacement, Reduction, and Refinement of animals).
Zebrafish possess several characteristics that make them an excellent NAM for studying immunotoxicity. Firstly, their immune system shares a remarkable degree of similarity with that of mammals, including humans (in overall, present around 70-75% of genetic homology with humans). Zebrafish possess functional counterparts of many immune cell types, including neutrophils, which play a crucial role in the innate immune response. The conservation of immune-related genes and pathways enables extrapolation of findings from zebrafish studies to human immune responses in an in vivo context. Secondly, zebrafish embryos are optically transparent, facilitating the visualization and tracking of immune cell behavior in real-time. This transparency allows researchers to label neutrophils or other immune cells with fluorescent markers and observe their migration patterns non-invasively under a microscope.
Understanding Immunotoxicity and Neutrophil Migration
The immune system plays a vital role in defending the body against foreign invaders, such as pathogens, and maintaining tissue homeostasis; therefore, it is vital for maintaining whole-body physiology. Immunotoxicity occurs when a chemical substance, such as a Drug or an environmental contaminant, disrupts the normal functioning of the immune system, leading to increased susceptibility to infections, autoimmunity, or other immune-related disorders.
The immune system is divided into innate and adaptative systems that are complementary to each other. The innate immune system acts rapidly through an unspecific response, while the adaptative immune system produces specific responses, much more effective but that take longer to be produced.
Neutrophils are the first responders to inflammation and infection, making them key players in the innate immune response. Their ability to migrate to the site of injury or infection is critical for initiating the immune defense, based on their phagocytic capacity and modulation of the immune system through the release of cytokines and chemokines. Dysregulation of neutrophil migration has been implicated in a wide range of immune-related disorders and evaluating this migration in response to potential immunotoxicants provides valuable insights into the compound's impact on immune function.
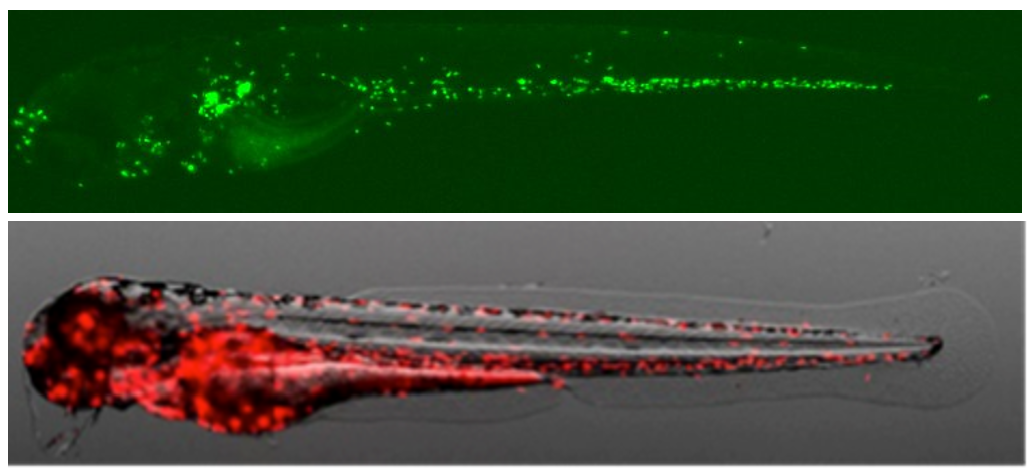
Zebrafish possess a sophisticated immune system, similar to that of humans, including an abundance of neutrophils. These cells exhibit conserved functions, such as phagocytosis, chemotaxis, and the ability to migrate toward sites of injury or infection. In this assay, researchers produce a specific stimulus, such as tissue damage, in the zebrafish larvae. The transparency of zebrafish embryos and larvae enables researchers to track individual neutrophils in real time and study their dynamic behavior during the immune response. They can visualize and quantify neutrophil migration by using fluorescent dyes or genetically encoded fluorescent proteins.
Advantages of Zebrafish Neutrophil Migration Assay for Immunotoxicity Evaluation
Using zebrafish as a NAM for immunotoxicity assays may help to test the toxicity of pharmacological, agrochemical, industrial, or cosmetic compounds to the immune system in a High-Content-Screening (HCS) manner and cost-efficient way. This is of special relevance in the Early Drug Discovery process where a high number of candidates have to be tested in a time and cost-efficient manner.
The assay involves exposing zebrafish embryos or larvae to various concentrations of test compounds and monitoring the migration of neutrophils in response to an inflammatory stimulus. Neutrophil migration patterns can be quantified, compared to control groups, and analyzed to determine any compound-induced alterations.
The advantages of zebrafish Neutrophil Migration Assay for immunotoxicity evaluation include the following:
- Conservation of Immune Response: many key components of the immune system, including involved cell types, signaling pathways, and molecular mechanisms are conserved between zebrafish and humans. Studying neutrophil migration in zebrafish provides valuable insights into the fundamental principles of immune response that can be applied to human toxicology and Drug Discovery.
- In vivo relevance: zebrafish Neutrophil Migration Assay allows researchers to observe the effects of compounds on neutrophil behaviour in a living organism, providing valuable in vivo
- Genetic Manipulation: zebrafish genome is well-characterized, and genetic manipulation techniques are highly developed. Researchers can create transgenic zebrafish lines that express fluorescent proteins exclusively in neutrophils or other immune cells, facilitating precise visualization and analysis of migration patterns.
- High-Content Screening (HCS): the zebrafish Neutrophil Migration Assay can be performed in a HCS manner, allowing the assessment of multiple compounds and concentraitons simultaneously. This accelerates the proper characterization of immunomodulator compounds and the early stages of Drug Development by rapidly identifying potential immunotoxicants and prioritizing safer candidates for further investigation. In addition, enables faster toxicological analysis for assessing immunotoxicity effects.
- Optical transparency and real-time visualization: zebrafish embryos are transparent, enabling non-invasive visualization of neutrophil migration in real time. Researchers can label neutrophils with fluorescent markers and track their movement under a microscope. This feature facilitates continuous monitoring of the immune response over time, offering dynamic insights into the compound's effects.
- Reduction in animal usage: the use of zebrafish for Neutrophil Migration Assays significantly reduces the number of experimental animals required. This reduction aligns with the principles of the 3Rs as the assay is performed in larvae of less than 6 days post-fertilization, before the animals enter under animal care policy surveillance.
Biobide’s Neutrophil Migration Assay
Biobide has developed a Neutrophil Migration Assay, a HCS assay for the identification of potential immunotoxic/immunosuppressant substances based on neutrophil migration in zebrafish embryos.
Sterile inflammation is induced by tail injury of transgenic embryos to evaluate neutrophil response to inflammation in the presence of test compounds. Wild-type zebrafish embryos are pre-incubated with different concentrations of the test item. Tail fins are amputated with a sterile scalpel or cryoinjury, and embryos are transferred to a 96-well plate. Images are taken at different time points post-wound and the number of neutrophils migrated to the tail wound are quantified. The decrease in the number of neutrophils migrated to the tail wound is related to a disruption of the immune response to the inflammation caused by the test compound.
In addition, this assay can be complemented with the Leucocyte Quantification Assay using a transgenic zebrafish expressing fluorescent neutrophils and macrophages, and developing quantitative PCRs of the main cytokines and chemokines.
Conclusion
A proper evaluation of disturbances of the immune system caused by chemical compounds is tremendously relevant as this system is a complex network of cells that work co-ordinately to ensure a proper defence and repair of the body and that can be affected by toxic compounds in many different ways.
Zebrafish Neutrophil Migration Assay offers a valuable and efficient approach for HCS of immunomodulation during the early stages of compound development and safety assessment. The use of zebrafish as a model organism provides a bridge between in vitro and in vivo mammalian models, combining the advantages of genetic conservation, optical transparency, and real-time visualization.
By leveraging this powerful tool, researchers can gain insights into compound-induced immunotoxic effects, contributing to safer, faster, and more effective Drug Discovery and Development processes and chemical safety evaluations.
Sources
- https://biobide.com/portfolio/immunotox-assay
- Immunotoxicity Testing Guidance | FDA
- https://www.ema.europa.eu/en/ich-s8-immunotoxicity-studies-human-pharmaceuticals-scientific-guideline
- Transgenic zebrafish (Danio rerio) as an emerging model system in ecotoxicology and toxicology: Historical review, recent advances, and trends - PubMed (nih.gov)
- Steamed Panax notoginseng and its Saponins Inhibit the Migration and Induce the Apoptosis of Neutrophils in a Zebrafish Tail-Fin Amputation Model - PubMed (nih.gov)
- Follow that cell: Leukocyte migration in L-plastin mutant zebrafish - PubMed (nih.gov)
- Exposure to perfluorooctanoic acid (PFOA) decreases neutrophil migration response to injury in zebrafish embryos - PubMed (nih.gov)
- A zebrafish compound screen reveals modulation of neutrophil reverse migration as an anti-inflammatory mechanism - PubMed (nih.gov)





